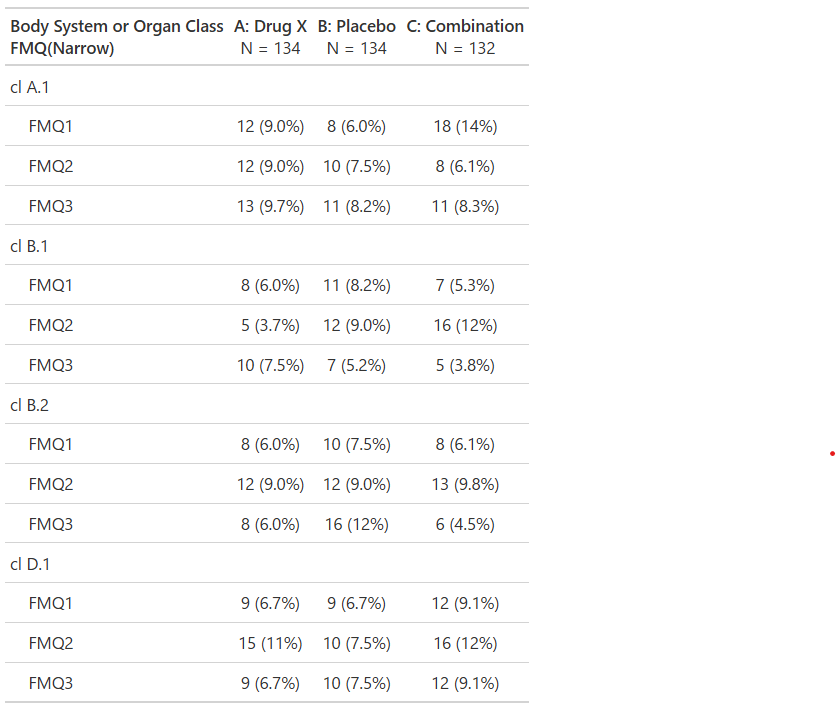
Subjects With Serious Adverse Events by System Organ Class and Preferred Term, Safety Population, Pooled Analysis (or Trial X)
FDA Table 10
table
FDA
safety
adverse events
Code
# Load libraries & data -------------------------------------
library(dplyr)
library(gtsummary)
adsl <- pharmaverseadam::adsl
adae <- pharmaverseadam::adae
# Pre-processing --------------------------------------------
adae <- adae |>
filter(
# safety population
SAFFL == "Y",
# serious adverse events
AESER == "Y"
)
$tbl_hierarchical{cards} data frame: 36 x 15 group1 group1_level group2 group2_level variable variable_level context stat_name stat_label stat stat_fmt fmt_fun warning error gts_column
1 <NA> <NA> TRT01A Placebo tabulate n n 86 86 0 stat_1
2 <NA> <NA> TRT01A Placebo tabulate N N 306 306 0 stat_1
3 <NA> <NA> TRT01A Placebo tabulate p % 0.281 28.1 <fn> stat_1
4 <NA> <NA> TRT01A Screen F… tabulate n n 52 52 0 stat_2
5 <NA> <NA> TRT01A Screen F… tabulate N N 306 306 0 stat_2
6 <NA> <NA> TRT01A Screen F… tabulate p % 0.17 17.0 <fn> stat_2
7 <NA> <NA> TRT01A Xanomeli… tabulate n n 72 72 0 stat_3
8 <NA> <NA> TRT01A Xanomeli… tabulate N N 306 306 0 stat_3
9 <NA> <NA> TRT01A Xanomeli… tabulate p % 0.235 23.5 <fn> stat_3
10 <NA> <NA> TRT01A Xanomeli… tabulate n n 96 96 0 stat_4ℹ 26 more rowsℹ Use `print(n = ...)` to see more rows